AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Bromine atomic radius1/10/2024 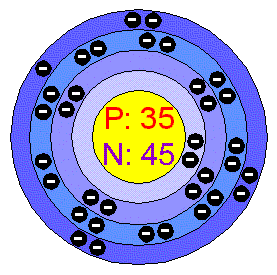
The French chemist and physicist Joseph-Louis Gay-Lussac suggested the name bromine due to the characteristic smell of the vapors.īromine occurs in nature as bromide salts in very diffuse amounts in crustal rock. With aniline, 2,4,6 tribromoaniline will precipitate (even in water)īromine was discovered by Antoine Balard at the salt marshes of Montpellier in 1826 but was not produced in quantity until 1860. When added to phenol a white precipitate, 2,4,6-tribromophenol, will form.For example, reaction with ethylene will produce 1,2-dibromoethane. When added to an alkene it will lose its colour as it reacts forming a colorless bromoalkane.It is also used to form intermediates in organic synthesis, where it is preferred to iodine due to its much lower cost.īromine is used to make brominated vegetable oil, which is used as an emulsifier in many citrus-flavored soft drinks.Īqueous bromine is orange and can be used in tests for alkenes and phenols. Traditionally the largest use of bromine was in the production of 1,2-dibromoethane which in turn was used as a gasoline anti- knock agent for leaded gasolines before they were largely phased out due to environmental considerations.īromine is also used in the manufacture of fumigants, brominated flame-retardants, water purification compounds, dyes, medicines, sanitizers, inorganic bromides for photography, etc. With many of the metals and elements, anhydrous bromine is less reactive than wet bromine however, dry bromine reacts vigorously with aluminium, titanium, mercury as well as alkaline earth metals and alkaline metals.ĭue to its contribution to ozone depletion in Earth's atmosphere, bromine has been evaluated to have an ozone depletion potential of 60 when compared to chlorine.Įlemental bromine is used to manufacture a wide variety of bromine compounds used in industry and agriculture. It reacts vigorously with amines, alkenes and phenols as well as aliphatic and aromatic hydrocarbons, ketones and acids (these are brominated by either addition or substitution reactions). It bonds easily with many elements and has a strong bleaching action.īromine is highly reactive and is a powerful oxidizing agent in the presence of water. 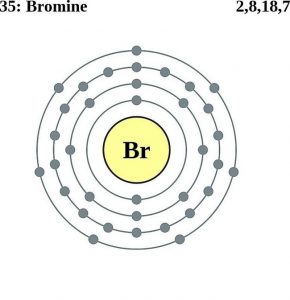
Bromine is slightly soluble in water, and highly soluble in carbon disulfide, aliphatic alcohols (such as methanol), and acetic acid. A halogen, bromine resembles chlorine chemically but is less active. It is a heavy, mobile, reddish-brown liquid, that evaporates easily at standard temperature and pressures in a red vapor (its colour resembles nitrogen dioxide) that has a strong disagreeable odour resembling that of chlorine. The pure chemical element has the physical form of a diatomic molecule, Br 2. Bromine vapors are very toxic upon inhalation.īromine is the only liquid nonmetallic element at room temperature and one of five elements on the period table that are liquid at or close to room temperature. This element is corrosive to human tissue in a liquid state and its vapors irritate eyes and throat. A halogen element, bromine is a red volatile liquid at standard room temperature which has a reactivity between chlorine and iodine. Bromine monochloride, a compound with formula BrCl, is employed in analytical chemistry and industrial cooling water systems.Bromine ( IPA: /ˈbrəʊmiːn/, Greek: βρωμος, brómos, meaning "stench"), is a chemical element in the periodic table that has the symbol Br and atomic number 35. Silver bromide, a compound with the formula AgBr, is mainly used in photographic films. Tetrabromoethane (C2H2Br4) and Bromoform (CHBr3) are used as liquids in gauges. It is also important in the production of inorganic and organic bromine alloys. 
Hydrogen bromide, a colorless compound with the formula HBr, is used as a catalyst in organic chemistry. Uses of Bromineīromine, a reddish-brown element with symbol Br, is used in making dyes, in pharmaceuticals, in flame-retardants, in medicines, and in agricultural chemicals. The compounds of this chemical element are added to various materials to reduce their flammability. This chemical element is used in chemical industry for producing insecticides, pesticides, sedatives, and other chemicals. In nature, it can be found in some deposits in the soils, which are in abundance in North America and China, as well as in the Dead Sea. Bromine belongs to the group of halogens. This chemical element exists in our cells in the form of bromide, but in large doses it has very strong irritating and toxic properties so it should be avoided. That is why its name comes from a Greek word meaning stench. Bromine is a toxic oily liquid of intense red color, known for its strong unpleasant smell.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
